National quality control laboratories (NQCL) for medicines play a vital role in protecting the public’s health by monitoring and verifying the quality and safety of pharmaceutical products in retail outlets, clinics, and hospitals. In addition to ensuring quality through testing, these laboratories alert regulators and manufacturers to take corrective action if needed.
In Ghana, the responsibility of medicines quality control resides with Ghana’s Food and Drugs Authority (FDA) Drug Physicochemical Laboratory and the Pharmaceutical Microbiology Laboratory within the Centre for Laboratory Services and Research.
Regional center of excellence
Thanks in part to ongoing support from the U.S. Agency for International Development (USAID) through its Promoting the Quality of Medicines Plus (PQM+) and predecessor programs, Ghana’s Drug Physicochemical Laboratory became the first World Health Organization-prequalified (WHO PQ) national medicines quality control laboratory in West Africa in 2022.
FDA Ghana has six laboratories that test drugs, medical devices, food, and cosmetics and household chemical substances each of which is internationally recognized through accreditation according to ISO/IEC 17025 standards:
- Drug physicochemical (DPLY)
- Pharmaceutical microbiology (PML)
- Medical devices
- Food microbiology
- Food physicochemical
- Cosmetic/household chemical substances
This accreditation signifies the lab can provide testing for U.N. agencies and international donors. Prequalification by WHO additionally indicates that a laboratory follows its Good Practices for Quality Control Laboratories, Good Practices for Pharmaceutical Microbiological Laboratories, or the relevant parts of WHO’s Good Manufacturing Practices (GMP) for chemical and microbiological analysis of pharmaceuticals and related products.

Physiochemical laboratory becomes a regional model
In August 2022, the Drug Physicochemical Laboratory achieved WHO PQ status for chemical testing of medicines, a feat it accomplished with no external help. This milestone is a critical step for Ghana, the region, and the African continent. As a result, the lab is now positioned to serve as a regional center of excellence to provide training to other medicines regulatory authorities (MRAs) and NQCLs in West Africa. FDA Ghana can also participate in international bids to analyze products procured by U.N. agencies, which would boost its financial independence.
With the largest accredited scope in Africa, FDA Ghana can be called a medicines quality testing powerhouse. This international recognition puts the FDA in a position to ensure country-wide and continental access to quality medicines as well as boost the status of Ghana-manufactured medicines in regional and national markets. It will also help to optimize limited health resources not only in Ghana, but also on the continent. Now with the WHO PQ of its Drug Physicochemical Laboratory, Ghana and other West African countries may no longer be required to ship samples to laboratories in Europe, spending precious time and money waiting for results, but rather can test them in country as this laboratory meets both of the international recognition (ISO/IEC 17025 and WHO PQ) standards required of testing laboratories.
Strong partnerships build sustainability
USAID has partnered with FDA Ghana for more than a decade to strengthen its medicines testing laboratories, beginning in 2004 under the Drug Quality and Information (DQI) program, which provided lab equipment and training. The PQM program began collaborating with FDA Ghana in 2011 by identifying critical gaps in the lab’s capacity to assess pharmaceutical quality and developing a roadmap toward achieving ISO/IEC 17025 and WHO PQ. PQM also evaluated the equipment, workspaces, staff skills, and their ability to generate reliable data. To address these gaps, PQM and FDA Ghana designed a multi-year, multi-layered approach to build the capacity and increase the sustainability of the lab.
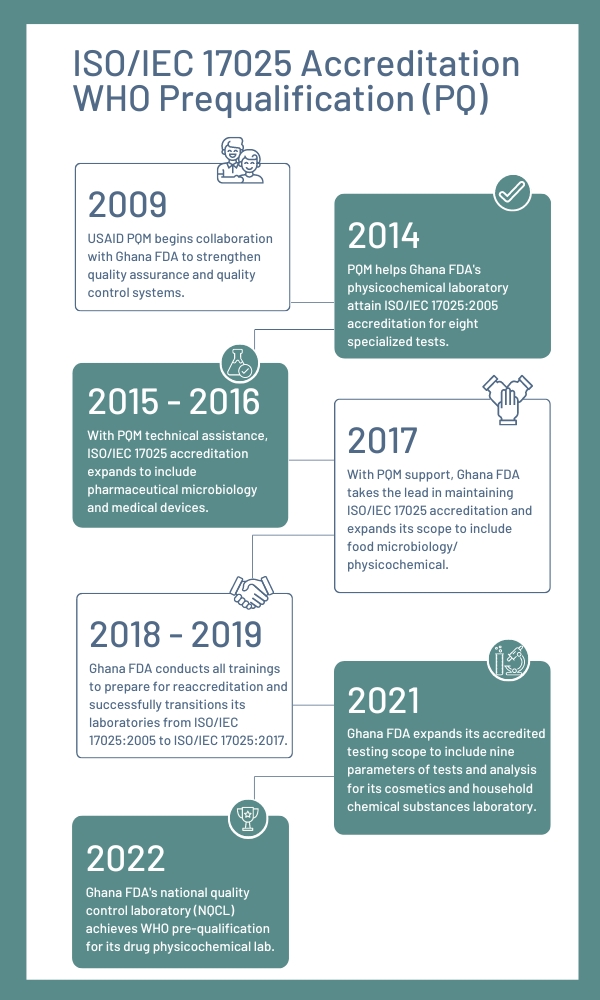
PQM was instrumental in identifying the test methods for accreditation, establishing priorities, building staff capacity, and developing an ISO/IEC 17025 implementation plan. Closing gaps in the quality management system was critical to obtaining the primary scope of accreditation.
USAID’s PQM+ program, which is led by USP, continues to support Ghana’s NQCL to build its sustainability and independence. As the only lab in the Economic Community of West African States (ECOWAS) region to receive WHO PQ, Ghana’s laboratory has become an important international player in the testing and verifying of finished pharmaceutical products.








